Different coatings
You will be aware that tablets can either be ‘uncoated’, ‘sugar coated’, ‘film coated’, ‘enteric coated’ or formulated to modify drug release (modified release). It is important to understand the reasons for the different tablet formulations before deciding whether it is safe or appropriate to recommending tablet crushing.
Each page explains the different concepts and the consequences of recommending that the formulation is crushed or placed in water to dissolve before swallowing.
Film and sugar coating
A sugar coating is basically is a thick, hard coating of sugar surrounding the tablet. It is no different in design to the sugar coatings placed on Smarties® or Minstrels ®. This is a traditional method used to hide the flavour of particularly unpleasant tasting drugs e.g. ibuprofen and quinine, both of which are very bitter. The other advantage of a sugar coating is that it can prevent light or moisture from affecting the drug’s stability.
Due to the increase in tablet size caused by sugar coating, drug manufacturers have largely changed to using ‘film coatings’. These are very thin layers of an inactive excipient coated thinly onto the tablet to again protect the tongue from the flavour of the contents and protect the contents from moisture and light. The film will break down with agitation from the stomach and significant amounts of moisture (saliva or stomach acid) and therefore does not significantly affect the way in which the drug is absorbed into the body.
Crushing these tablets therefore may not seriously effect how the drug is released but may cause the resultant mixture to be unpleasant to taste.
Enteric coating
If a tablet is described as having an ‘enteric coating’ (e/c) or ‘gastro-resistant’ it means that there is a coating which is designed to hold the tablet together in acid conditions i.e. the stomach and therefore release the drug in the intestines.
There are three reasons for putting such a coating on a tablet or capsule ingredient:
- To protect the stomach from the drug
- To protect the drug from the stomach
- To release the drug after the stomach e.g. in the intestines
The drugs which most commonly cause stomach ulcers like aspirin, diclofenac and naproxen are frequently available with enteric coatings. Omeprazole is itself broken down in acid and therefore the drug generally has an enteric coating around it either as a granule in the capsules or as a granule in the dispersible form. Sulfasalazine is used either for the treatment of arthritis or for the treatment of Crohn’s disease which is inflammation of the intestines. When used for arthritis, it is often given without an enteric coating so that it can be absorbed more quickly. For Crohn’s, it is needed to work in the intestines and it is given with an enteric coating.
It can be seen that an enteric coating is therefore advantageous and such tablets or the contents of enteric coated capsules should never be crushed before being taken.
Modified release
Modified release means that the escape of the drug from the tablet has been modified in some way. Usually this is to slow the release of the drug so that the medicine does not have to be taken too often and therefore improves compliance. The other benefit from modifying release is that the drug release is controlled and there are smaller peaks and troughs in blood levels therefore reducing the chance of peak effects and increasing the likelihood of therapeutic effectiveness for longer periods of time.
Tablets and capsules which are designed to provide modified release often have the letters MR, LA, XL, CR or SR in their names e.g. Diffundox MR, Elantan LA, Dilzem XL Calcicard CR, Dilcardia S. Sometimes the words ‘slow’ or ‘retard’ can be used to denote modified release e.g. Diclomax retard, Voltarol retard & Slow K.
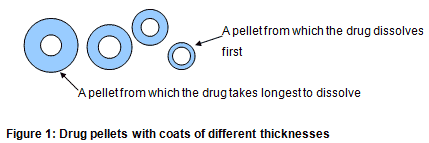
There are a number of ways in which a medicine can have its release modified. Perhaps the most famous is that used in Contac 400 capsules. The pellets inside are of different thicknesses and therefore the thinnest release the drug first and the thickest last (Figure 1).
Another method used is to put the drug in a viscous liquid which breaks down slowly itself and therefore releases the drug slowly. One method which has been tried in the past has been to put a non-dissolving coating around the tablet or capsule, laser a small hole in it and then let the drug only release through the hole. In such cases patients frequently report passing the tablet or capsule whole and worrying whether it is actually working. Frequently, they are just passing the outer coat in their stools as this is how the medicine is designed to work and the drug has long been absorbed.
Modified release products usually have a higher than normal amount of the drug within them and therefore if they are crushed the whole dose will be released very quickly and could be dangerous.
For this reason, modified release products should never be crushed or modified before being taken.